
F0 VALUE CALCULATION
The SAL value (Sterility Assurance Level) defines as the probability that viable microorganisms remain on the sterilized product after sterilization. According to the Ph.Eur., sterility is defined as an SAL value of 10-6. The temperature and duration of the sterilization process are the crucial parameters to determine whether the sterilized product achieves the desired SAL value. In steam sterilization, temperature values of 121 °C or 134°C have been established as standard.
The F0 value refers to the duration of the sterilization program, which can be determined by relying on the performance data of a highly heat-resistant bacterium, Geobacillus stearothermophilus, in a worst-case scenario. It is assumed that this bacterium is more stable than other microorganisms, and therefore, reaching the correct SAL for this one means reaching it for the other microorganisms present on the sterilized product as well. Thus, the data of Geobacillus stearothermophilus can be used to calculate the duration of the sterilization program. The D-value is also required, which indicates the time required to reduce the number of microorganisms by one logarithmic unit at a specific temperature. This value must be determined experimentally.
For the sterilization of waste, an overkill method is recommended, In this method, the sterilization is performed for a longer duration than indicated by these calculations. In the classical approach for highly contaminated goods, the sterilization time is doubled to achieve a reduction from 106 viable microorganisms to 10-6 viable microorganisms, to counteract possible fluctuations in temperature and exposure times within the waste, and to ensure a safe and reproducible sterilization result.
Then again, there are scenarios where the sterilization time should be minimized at a given temperature, for example, for sterilizing temperature-sensitive products or to increase production efficiency. In this case, the heating time from 100 °C to 121 °C and the cooling time can be included in the calculation. A temperature above 100 °C already has a detrimental effect on microorganisms and therefore has its own D-value (see figure). By considering this effect during the heating and cooling time, the holding time can be shortened accordingly. This allows the desired SAL value to be achieved with a shorter overall process duration. This must be done individually for each sterilizer, as the calculation should refer to the “coldest or most difficult-to-sterilize point” (European Pharmacopeia) in the load.
For less temperature-sensitive products, the duration of the sterilization process for solid materials can also be reduced by using higher temperatures for sterilization (typically 134 °C). This is where the Z-value comes into play, which describes the temperature increase that reduces the D-value by one tenth. Like the D-value, the Z-value is characteristic for each microorganism and is given in degrees Celsius.
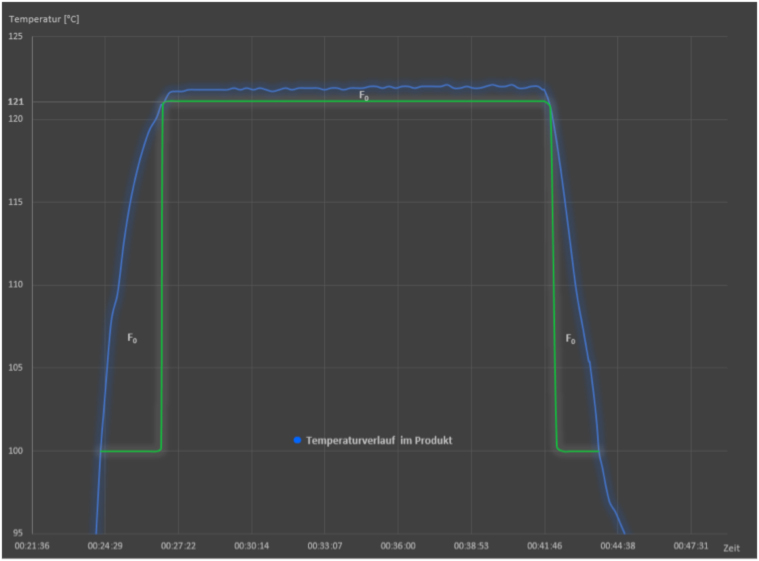
Graphical illustration of the inclusion of heating and cooling time for F0 value calculation. Blue: Temperature curve during sterilization in a steam sterilizer, holding time at ≥ 121 °C; green: D values used: The D values of 100 °C are used in the heating time and cooling time, and those of 121 °C in the holding time.
